Medical & Health Packaging IML Label Solutions
One-line Definition
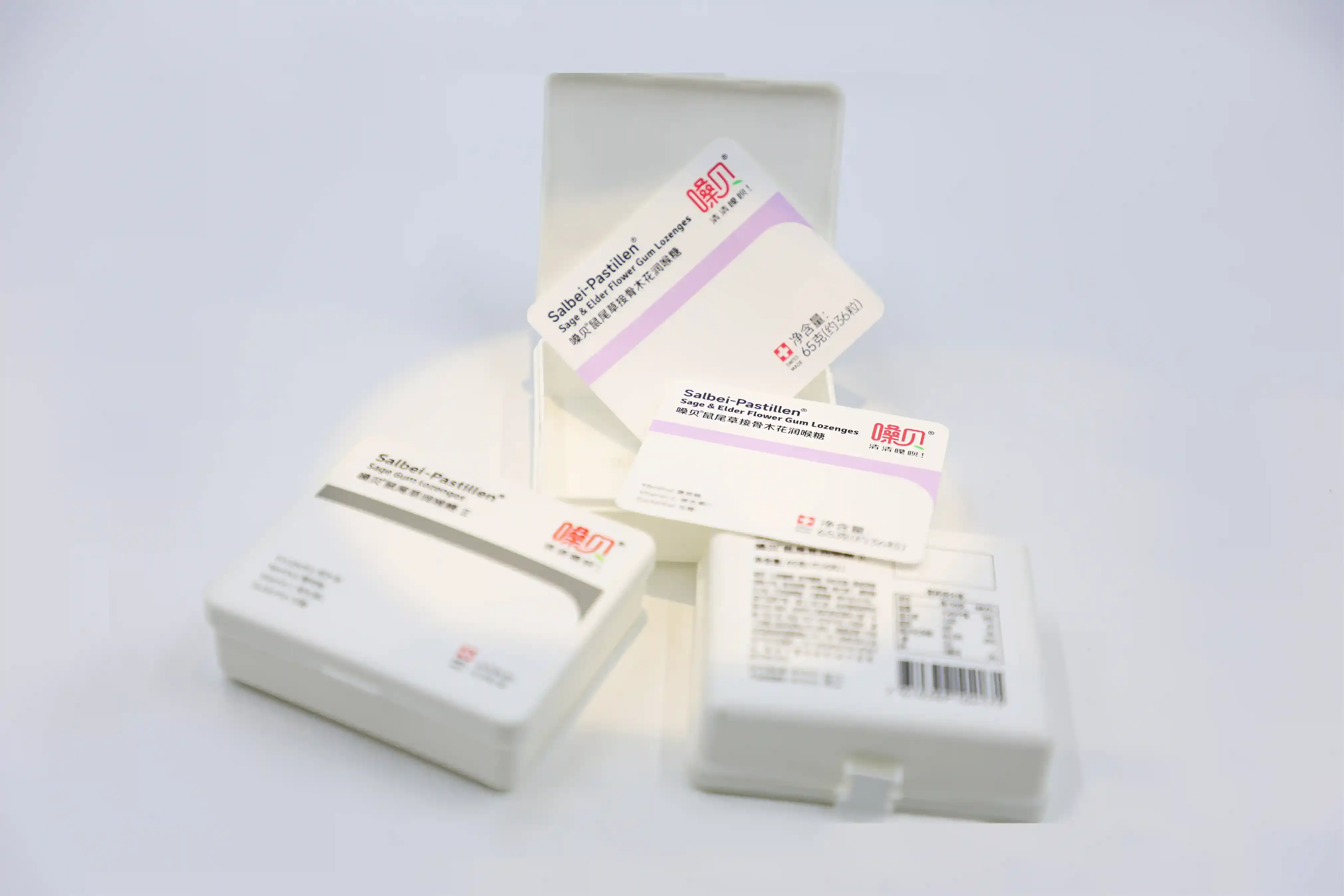
IML labeling solutions are developed for medical and health packaging with high cleanliness and stability requirements.
What It Is
In-Mold Labels fused with medical and health plastic packaging during molding, forming a durable and contamination-resistant surface.
Why It Matters
Medical and health products demand clean labeling, long-term legibility, and reduced contamination risk—areas where traditional labels may fail.
Material Type
- Low-migration ink systems
Surface Finish
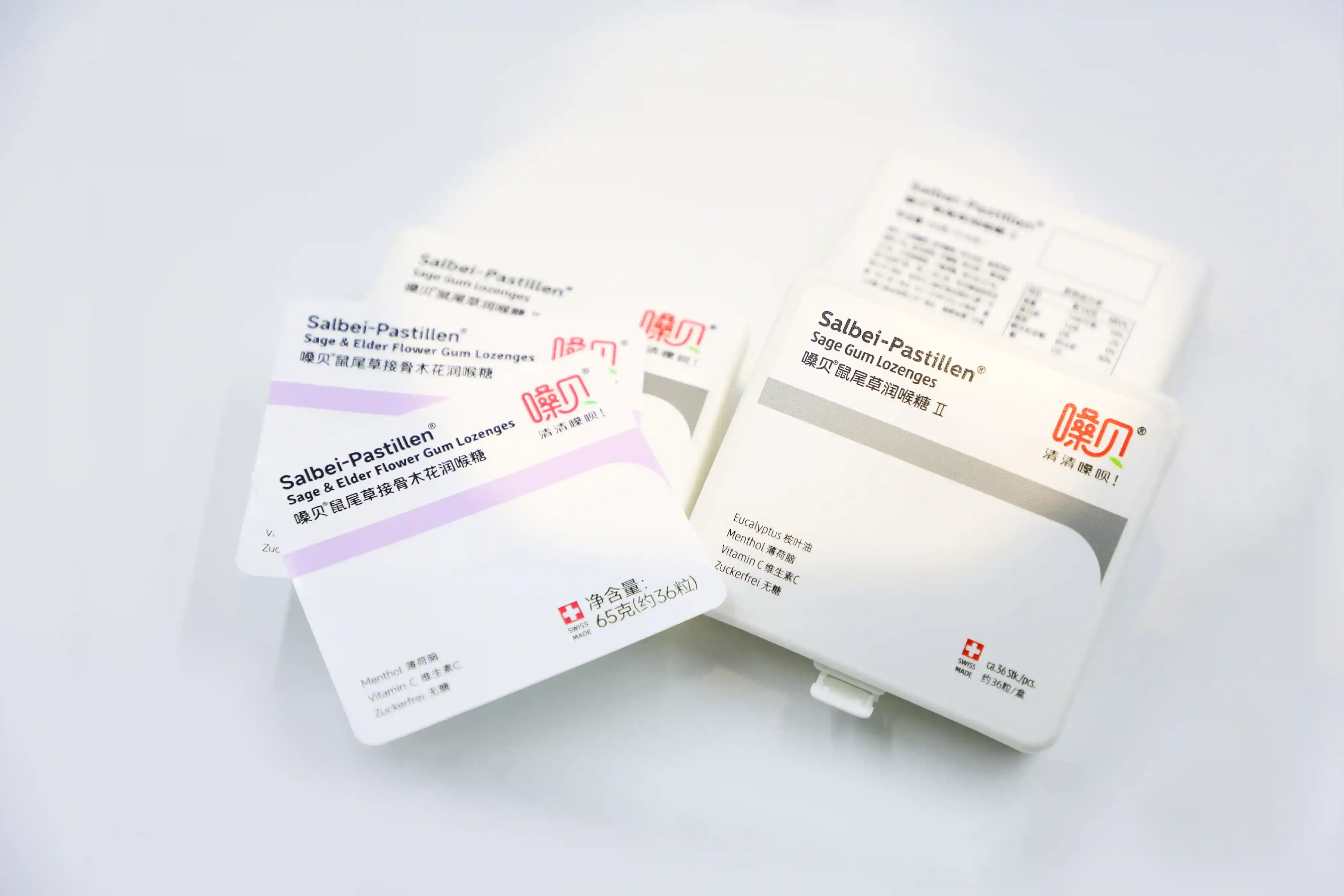
Printing Process
Structure Description
Multi-layer IML construction is designed for stability and abrasion resistance.
Thickness Range
Typically 50–70 μm
Compatibility
- Medical-grade plastic containers
Performance Highlights
- Reduced label separation risk
Typical Applications
- Pharmaceutical containers
- Medical disposables packaging
Target Products
- Health supplement containers
QC Control Points
ERP Traceability
Material batch and production lot tracking
Risk Prevention Notes
Validation testing recommended for regulated supply chains.
Hero Image
Clean, minimal medical packaging with IML decoration
Detail Image
Close-up of print clarity and surface texture
Application Image
Clinical or laboratory environment (equipment-focused)
Related By Industry
Related By Process
- 🔗 Quality Control in IML Printing
Related Materials Pages
- 🔗 Low-Migration Ink Systems
FAQ Q1
Is IML suitable for medical packaging?
FAQ A1
IML is suitable for non-sterile medical and health packaging where durability and label stability are required.
FAQ Q2
Can labeling remain legible over long storage periods?
FAQ A2
Yes. IML labels maintain print clarity and adhesion throughout the product lifecycle.
Primary CTA
Discuss Medical Packaging Requirements
Secondary CTA
Request Sample Labels
Contact Path
Technical Inquiry Form